|
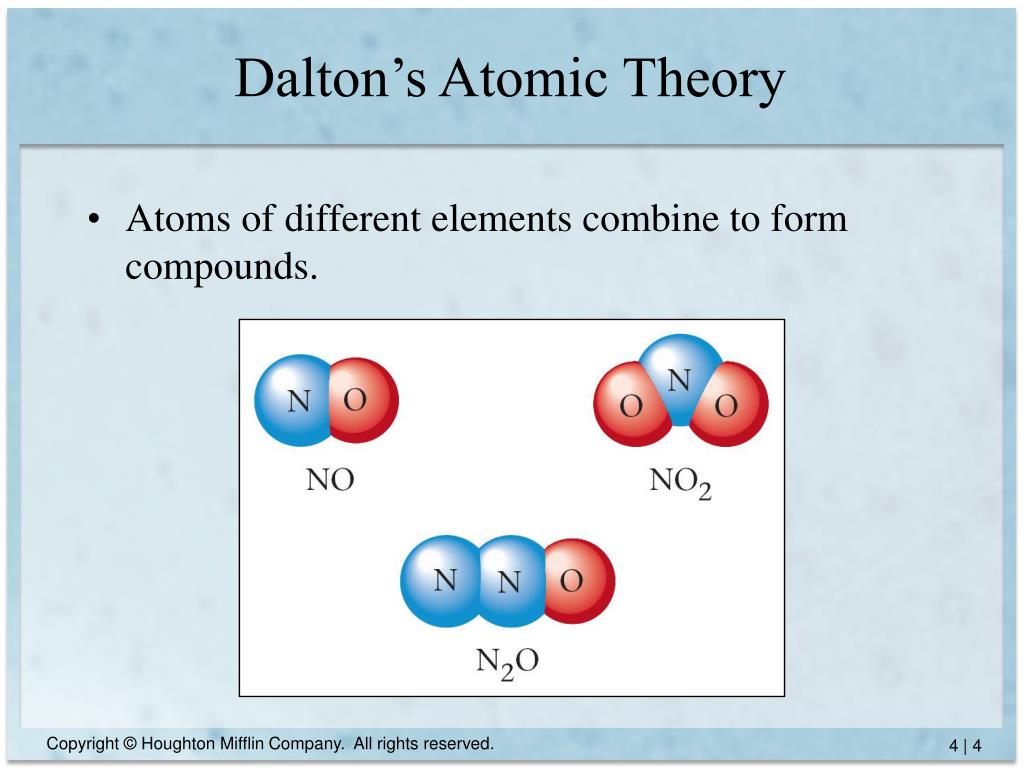
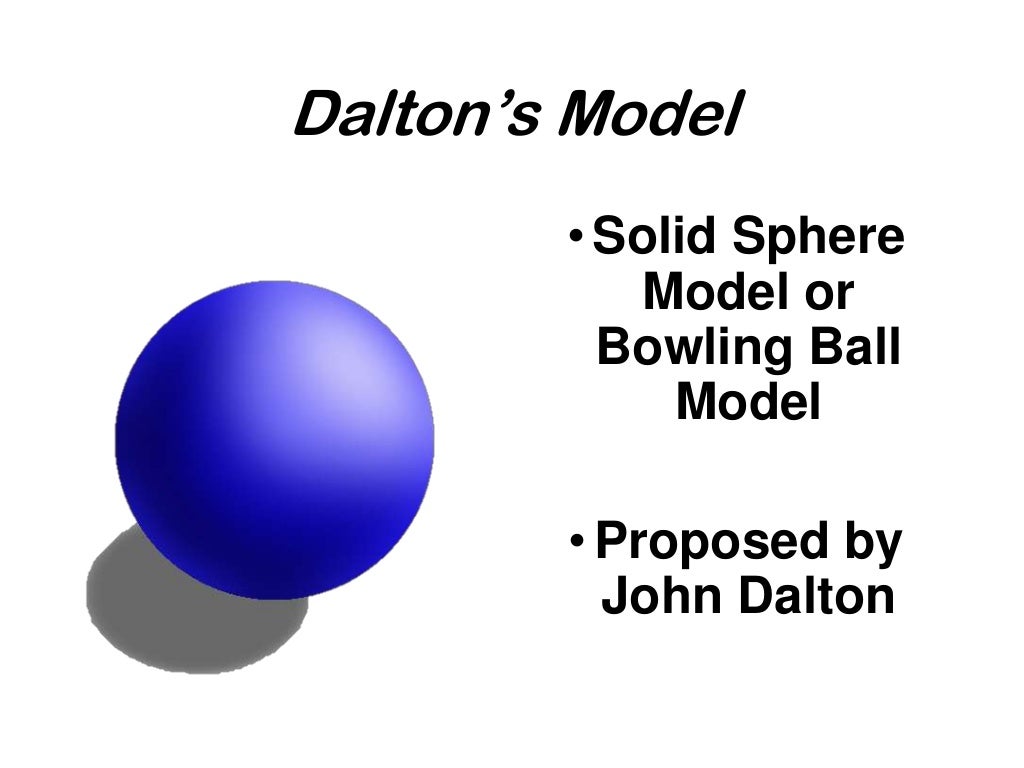
4/5/2024 0 Comments Dalton's atomic theory modelThere is a large difference between Dalton’s atomic theory and modern atomic theory since the structure and properties of atom according to Dalton’s theory is different from the structure and properties proposed by the modern atomic theory. This led to the development of modern atomic theory, which can explain almost everything about the structure and properties of atoms. Summary – Dalton’s Atomic Theory vs Modern Atomic TheoryĮven though there were no well-equipped laboratories, Dalton was able to build up a theory on atoms, which are invisible to the eye. Modern atomic theory states that subatomic particles can participate in reactions. Modern atomic theory explains the location, reactions, and behavior of electrons.ĭalton’s atomic theory explains that atoms are the smallest particle that can be engaged in reactions. Modern atomic theory explains details about isotopes having a different number of neutrons and the same number of protons.ĭalton couldn’t give details about electrons. It states that all atoms of the same element are identical. Modern atomic theory says that atoms are composed of subatomic particles protons, electrons, and neutrons.ĭalton’s theory does not explain details about isotopes. Modern atomic theory is the theory that explains the fully detailed structure of an atom.Īccording to Dalton’s atomic theory, atoms are indivisible particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
---teachoo-01.jpg)

 RSS Feed
RSS Feed
